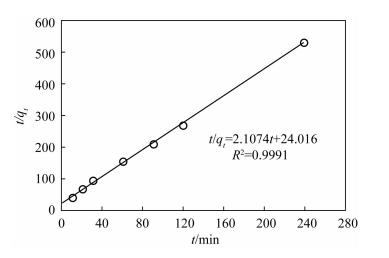
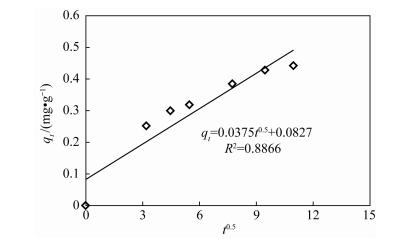
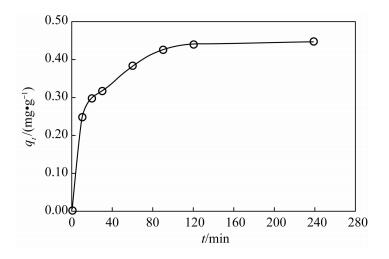
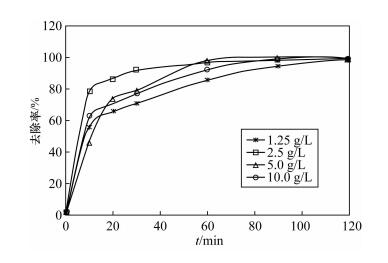
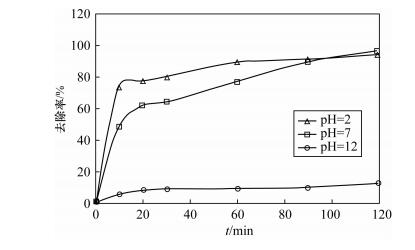
| [1] |
Mohan D, Pittman Jr C U.Arsenic removal from water/wastewater using adsorbents—A critical review [J].Journal of Hazardous Materials,2007, 142:1-53. doi: 10.1016/j.jhazmat.2007.01.006
CrossRef Google Scholar
|
| [2] |
Nickson R, McArthur J, Burgess W, Ahmed K M, Ravenscroft P, Rahman M.Arsenic poisoning of Bangladesh groundwater [J].Nature, 1998, 385:338.
Google Scholar
|
| [3] |
Watkims C D, de Groot P H.A perspective on the FOCUS Conference on Eastern regional ground water issues [J].Ground Water Management, 1991, 7:967-978.
Google Scholar
|
| [4] |
Das D, Chatterjee A, Mandal B K, Samanta G, Chakraborti D, Chanda B.Arsenic in ground water in six districts of West Bengal, India:The biggest arsenic callamity in the word (Part 2). Arsenic concentration in drinking water, hair, nails, urine, skin-scale, and liver tissue (biopsy) of the affected people [J].Analyst, 1995, 120:917-924. doi: 10.1039/an9952000917
CrossRef Google Scholar
|
| [5] |
肖唐付,洪冰,杨中华,杨帆.砷的水地球化学及环境效应[J].地质科技情报,2001,20(1):71-76.
Google Scholar
|
| [6] |
庄金陵.砷对世界地下水源的污染[J].矿产与地质, 2003, 17(2):177-178.
Google Scholar
|
| [7] |
Hering J G, Chen P Y, Wilkie J A, Elimelech M.Arsenic removal from drinking water during coagulation[J].Journal of Environment Engineering, 1997, 123(8):800-807. doi: 10.1061/(ASCE)0733-9372(1997)123:8(800)
CrossRef Google Scholar
|
| [8] |
Borho M, Wilderer P.Optimized removal of arsenate(Ⅲ) by adaptation of oxidation and precipitation processes to the filtration step [J].Water Science Technology, 1996, 34(9):25-31.
Google Scholar
|
| [9] |
苑宝玲,李坤林,邓临莉,张之东.多功能高铁酸盐去除饮用水中砷的研究[J].环境科学, 2006, 27(2):281-284.
Google Scholar
|
| [10] |
Daus B, Wennrich R, Weiss H.Sorption materials for arsenic removal from water:A comparative study [J].Water Research, 2004, 38:2948-2954. doi: 10.1016/j.watres.2004.04.003
CrossRef Google Scholar
|
| [11] |
Boddu V M, Abburi K, Talbott J L, Smith E D, Haasch R.Removal of arsenic(Ⅲ) and arsenic(Ⅴ) from aqueous medium using chitosan-coated biosorbent [J].Water Research, 2008, 42:633-642. doi: 10.1016/j.watres.2007.08.014
CrossRef Google Scholar
|
| [12] |
Jaeshin K, Benjamin M M.Modeling a novel ion exchange process for arsenic and nitrate removal [J].Water Research, 2004, 38:2053-2062. doi: 10.1016/j.watres.2004.01.012
CrossRef Google Scholar
|
| [13] |
Sato Y, Kang M, Kamei T, Magara Y.Performance of nanofiltration for arsenic removal[J].Water Research, 2002, 36:3371-3377. doi: 10.1016/S0043-1354(02)00037-4
CrossRef Google Scholar
|
| [14] |
van der Bruggen B, Vandecasteele C.Removal of pollutants from surface water and groundwater by nanofiltration:Overview of possible applications in the drinking water industry [J].Environment Pollution, 2003, 122:435-445. doi: 10.1016/S0269-7491(02)00308-1
CrossRef Google Scholar
|
| [15] |
Gholami M M, Mokhtari M A, Aameri A, Alizadeh F M R.Application of reverse osmosis technology for arsenic removal from drinking water [J].Desalination, 2006, 200(1-3):725-727. doi: 10.1016/j.desal.2006.03.504
CrossRef Google Scholar
|
| [16] |
Zouboulis A I, Katsoyiannis I A.Recent advances in the bioremediation of arsenic-contaminated groundwaters [J].Environment International, 2005, 31:213-219. doi: 10.1016/j.envint.2004.09.018
CrossRef Google Scholar
|
| [17] |
Al Rmalli S W, Harrington C F, Ayub M, Haris P I.A biomaterial based approach for arsenic removal from water [J].Journal Environment Monitoring,2005, 7:279-282. doi: 10.1039/b500932d
CrossRef Google Scholar
|
| [18] |
Mondal P, Majumder C B, Mohanty B.Removal of trivalent arsenic [As(Ⅲ)] from contaminated water by calcium chloride (CaCl2)-impregnated rice husk carbon[J].Industrial and Engineering Chemistry Research,2007, 46:2550-2557. doi: 10.1021/ie060702i
CrossRef Google Scholar
|
| [19] |
Li Z, Beachner R, McManama Z, Hanlie H.Sorption of arsenic by surfactant modified zeolite and kaolinite [J].Microporous Materials,2007, 105:291-297. doi: 10.1016/j.micromeso.2007.03.038
CrossRef Google Scholar
|
| [20] |
Streat M, Hellgardt K, Newton N L R.Hydrous ferric oxide as an adsorbent in water treatment. Part 2. Adsorption studies [J].Process Safety Environmental Protection,2008, 86:11-20. doi: 10.1016/j.psep.2007.10.008
CrossRef Google Scholar
|
| [21] |
Guo H, Stüben D, Berner Z.Adsorption of arsenic(Ⅲ) and arsenic(Ⅴ) from groundwater using natural siderite as the adsorbent[J].Journal of Colloid Interface Science,2007, 315:47-53. doi: 10.1016/j.jcis.2007.06.035
CrossRef Google Scholar
|
| [22] |
汪大翠,徐新华,宋爽.工业废水中专项污染物处理手册[M].北京:化学工业出版社,2000:66-79.
Google Scholar
|
| [23] |
杨杰,顾海红,赵浩,徐炎华.含砷废水处理技术研究进展[J].工业水处理,2003,23(6):14-18. doi: 10.11894/1005-829x.2003.23(6).14
CrossRef Google Scholar
|
| [24] |
黄园英,秦臻,刘菲.纳米铁去除饮用水中As(Ⅲ)和As(Ⅴ)[J].岩矿测试,2009,28(6):529-534.
Google Scholar
|
| [25] |
黄园英,刘丹丹,刘菲.纳米铁用于饮用水中As(Ⅲ)去除效果[J].生态环境学报,2009,18(1):83-87.
Google Scholar
|
| [26] |
朱慧杰,贾永峰,吴星,王赫.负载型纳米铁吸附剂去除饮用水中As(Ⅲ)的研究[J].环境科学,2009, 30(6):1644-1648.
Google Scholar
|
| [27] |
Boronina T N, Lagadic I, Sergeev G B, Klabunde K J.Activated and nonactivated forms of zinc powder:Reactivity toward chlorocarbons in water and AFM studies of surface morphologies[J].Environmental Science & Technology,1998, 32:2614-2622.
Google Scholar
|
| [28] |
Choia J H, Kim Y H.Reduction of 2,4,6-trichlorophenol with zero-valent zinc and catalyzed zinc [J].Journal of Hazardous Materials, 2009, 166:984-991. doi: 10.1016/j.jhazmat.2008.12.004
CrossRef Google Scholar
|
| [29] |
Roberts A L, Totten L A, Arnold W A, Burris D R, Campbell T J.Reductive elimination of chlorinated ethylenes by zero-valent metals [J].Environmental Science & Technology,1996,30:2654-2659.
Google Scholar
|
| [30] |
Arnold W A, Roberts A L.Pathways of chlorinated ethylene and chlorinated acetylene reaction with Zn(0)[J].Environmental Science & Technology,1998, 32:3017-3025.
Google Scholar
|
| [31] |
Fennelly J P, Roberts A L.Reaction of 1,1,1-trichloroethane with zero-valent metals and bimetallic reductants[J].Environmental Science & Technology, 1998, 32:1980-1988.
Google Scholar
|
| [32] |
Ho Y S.Citation review of Lagergren kinetic rate equation on adsorption reactions [J].Scientometrics, 2004, 59:171-177. doi: 10.1023/B:SCIE.0000013305.99473.cf
CrossRef Google Scholar
|
| [33] |
Azizian S.Kinetic models of sorption:A theoretical analysis [J].Journal of Colloid Interface Science, 2004, 276:47-52. doi: 10.1016/j.jcis.2004.03.048
CrossRef Google Scholar
|
| [34] |
Lagergren S.About the theory of so-called adsorption of soluble substance [J].Kungliga Svenska Vetenskap-sakademiens Handlingar, 1898, 24(4):1-39.
Google Scholar
|
| [35] |
Ho Y S.Review of second-order models for adsorption systems [J].Journal of Hazardous Materials, 2006, 136:681-689. doi: 10.1016/j.jhazmat.2005.12.043
CrossRef Google Scholar
|
| [36] |
Borah D, Satokawa S, Kato S, Kojima T.Sorption of As(Ⅴ) from aqueous solution using acid modified carbon black [J].Journal of Hazardous Materials, 2009,162:1269-1277. doi: 10.1016/j.jhazmat.2008.06.015
CrossRef Google Scholar
|
| [37] |
Ozdes D, Gundogdu A, Kemer B, Duran C, Senturk H B, Soylak M. Removal of Pb(Ⅱ) ions from aqueous solution by a waste mud from copper mine industry:Equilibrium, kinetic and thermodynamic study [J].Journal of Hazardous Materials,2009, 166:1480-1487. doi: 10.1016/j.jhazmat.2008.12.073
CrossRef Google Scholar
|
| [38] |
Reed B E, Vaughan R, Jiang L. As(Ⅲ), As(Ⅴ), Hg and Pb removal by Fe-oxide impregnated activated carbon [J]. Journal of Environment Engineering, 2000, 126: 869-873. doi: 10.1061/(ASCE)0733-9372(2000)126:9(869)
CrossRef Google Scholar
|
| [39] |
Mondal P, Balomajumder C, Mohanty B.A laboratory study for the treatment of arsenic, iron, and manganese bearing ground water using Fe3+ impregnated activated carbon:Effects of shaking time, pH and temperature [J]. Journal of Hazardous Materials, 2007, 144:420-426. doi: 10.1016/j.jhazmat.2006.10.078
CrossRef Google Scholar
|
| [40] |
杨力.砷污染及含砷废水治理[J].有色金属加工,1999(4):27-29.
Google Scholar
|
| [41] |
Pattanayak J, Mondal K, Mathew S, Lalvani S B.A paraletric evaluation of the removal of As(Ⅴ) and As(Ⅲ) by carbon-based adsorbents [J].Carbon, 2000, 38(4):589-596. doi: 10.1016/S0008-6223(99)00144-X
CrossRef Google Scholar
|
| [42] |
Ning R Y.Arsefiic removal by reverse osnosis [J].Desalination, 2002,143(3):237-241. doi: 10.1016/S0011-9164(02)00262-X
CrossRef Google Scholar
|





 DownLoad:
DownLoad: