| Citation: | Zheng-cai Zhang, Neng-you Wu, Chang-ling Liu, Xi-luo Hao, Yong-chao Zhang, Kai Gao, Bo Peng, Chao Zheng, Wei Tang, Guang-jun Guo, 2022. Molecular simulation studies on natural gas hydrates nucleation and growth: A review, China Geology, 5, 330-344. doi: 10.31035/cg2022017 |
Molecular simulation studies on natural gas hydrates nucleation and growth: A review
-
Abstract
How natural gas hydrates nucleate and grow is a crucial scientific question. The research on it will help solve practical problems encountered in hydrate accumulation, development, and utilization of hydrate related technology. Due to its limitations on both spatial and temporal dimensions, experiment cannot fully explain this issue on a micro-scale. With the development of computer technology, molecular simulation has been widely used in the study of hydrate formation because it can observe the nucleation and growth process of hydrates at the molecular level. This review will assess the recent progresses in molecular dynamics simulation of hydrate nucleation and growth, as well as the enlightening significance of these developments in hydrate applications. At the same time, combined with the problems encountered in recent hydrate trial mining and applications, some potential directions for molecular simulation in the research of hydrate nucleation and growth are proposed, and the future of molecular simulation research on hydrate nucleation and growth is prospected.
-
-
References
Abascal JLF, Sanz E, García Fernández R, Vega C. 2005. A potential model for the study of ices and amorphous water: TIP4P/Ice. Journal of Chemical Physics, 122(23), 234511. doi: 10.1063/1.1931662. Abascal JLF, Vega C. 2005. A general purpose model for the condensed phases of water: TIP4P/2005. Journal of Chemical Physics, 123(23), 234505. doi: 10.1063/1.2121687. Andreassen K, Hubbard A, Winsborrow M, Patton H, Vadakkepuliyambatta S, Plaza-Faverola A, Gudlaugsson E, Serov P, Deryabin A, Mattingsdal R, Mienert J, Bunz S. 2017. Massive blow-out craters formed by hydrate-controlled methane expulsion from the Arctic seafloor. Science, 356(6341), 948–953. doi: 10.1126/science.aal4500. Arjun, Berendsen TA, Bolhuis PG. 2019. Unbiased atomistic insight in the competing nucleation mechanisms of methane hydrates. Proceedings of the National Academy of Sciences of the United States of America, 116(39), 19305–19310. doi: 10.1073/pnas.1906502116. Arjun A, Bolhuis PG. 2020. Rate prediction for homogeneous nucleation of methane hydrate at moderate supersaturation using transition interface sampling. The Journal of Physical Chemistry B, 124(37), 8099–8109. doi: 10.1021/acs.jpcb.0c04582. Arjun A, Bolhuis PG. 2021. Homogenous nucleation rate of CO2 hydrates using transition interface sampling. Journal of Chemical Physics, 154(16), 164507. doi: 10.1063/5.0044883. Bai DS, Chen GJ, Zhang XR, Wang WC. 2011. Microsecond molecular dynamics simulations of the kinetic pathways of gas hydrate formation from solid surfaces. Langmuir, 27(10), 5961–5967. doi: 10.1021/la105088b. Bai DS, Chen GJ, Zhang XR, Wang WC. 2012. Nucleation of the CO2 hydrate from three-phase contact lines. Langmuir, 28(20), 7730–7736. doi: 10.1021/la300647s. Bai DS, Chen GJ, Zhang XR, Sum AK, Wang WC. 2015a. How properties of solid surfaces modulate the nucleation of gas hydrate. Scientific Reports, 5, 12747. doi: 10.1038/srep12747. Bai DS, Zhang DW, Zhang XR, Chen GJ. 2015b. Origin of self-preservation effect for hydrate decomposition: Coupling of mass and heat transfer resistances. Scientific Reports, 5, 14599. doi: 10.1038/srep14599. Barnes BC, Knott BC, Beckham GT, Wu DT, Sum AK. 2014. Reaction coordinate of incipient methane clathrate hydrate nucleation. The Journal of Physical Chemistry B, 118(46), 13236–13243. doi: 10.1021/jp507959q. Bellucci MA, Walsh MR, Trout BL. 2018. Molecular dynamics analysis of anti-agglomerant surface adsorption in natural gas hydrates. The Journal of Physical Chemistry C, 122(5), 2673–2683. doi: 10.1021/acs.jpcc.7b09573. Bi YF, Li TS. 2014. Probing methane hydrate nucleation through the forward flux sampling method. The Journal of Physical Chemistry B, 118(47), 13324–13332. doi: 10.1021/jp503000u. Bi YF, Porras A, Li TS. 2016. Free energy landscape and molecular pathways of gas hydrate nucleation. Journal of Chemical Physics, 145(21), 211909. doi: 10.1063/1.4961241. Bui T, Sicard F, Monteiro D, Lan Q, Ceglio M, Burress C, Striolo A. 2018. Antiagglomerants affect gas hydrate growth. The Journal of Physical Chemistry Letters, 9(12), 3491–3496. doi: 10.1021/acs.jpclett.8b01180. Bui T, Monteiro D, Vo L, Striolo A. 2020. Synergistic and antagonistic effects of aromatics on the agglomeration of gas hydrates. Scientific Reports, 10(1), 5496. doi: 10.1038/s41598-020-62060-5. Cao PQ, Sheng JL, Wu JY, Ning FL. 2021. Mechanical creep instability of nanocrystalline methane hydrates. Physical Chemistry Chemical Physics, 23(5), 3615–3626. doi: 10.1039/d0cp05896c. Chan H, Narayanan B, Cherukara MJ, Sen FG, Sasikumar K, Gray SK, Chan MKY, Sankaranarayanan SKRS. 2019. Machine learning classical interatomic potentials for molecular dynamics from first-principles training data. The Journal of Physical Chemistry C, 123(12), 6941–6957. doi: 10.1021/acs.jpcc.8b09917. Chen Y, Chen C, Sum AK. 2021. Molecular resolution into the nucleation and crystal growth of clathrate hydrates formed from methane and propane mixtures. Crystal Growth & Design, 21(2), 960–973. doi: 10.1021/acs.cgd.0c01303. Christiansen RL, Sloan ED. 1994. Mechanisms and kinetics of hydrate formation. Annals of the New York Academy of Sciences, 715(1), 283–305. doi: 10.1111/j.1749-6632.1994.tb38841.x. Cox SJ, Taylor DJF, Youngs TGA, Soper AK, Totton TS, Chapman RG, Arjmandi M, Hodges MG, Skipper NT, Michaelides A. 2018. Formation of methane hydrate in the presence of natural and synthetic nanoparticles. Journal of the American Chemical Society, 140(9), 3277–3284. doi: 10.1021/jacs.7b12050. Dec SF, Bowler KE, Stadterman LL, Koh CA, Sloan ED, Jr. 2006. Direct measure of the hydration number of aqueous methane. Journal of the American Chemical Society, 128(2), 414–415. doi: 10.1021/ja055283f. English NJ, MacElroy JMD. 2015. Perspectives on molecular simulation of clathrate hydrates: Progress, prospects and challenges. Chemical Engineering Science, 121, 133–156. doi: 10.1016/j.ces.2014.07.047. Falenty A, Hansen TC, Kuhs WF. 2014. Formation and properties of ice XVI obtained by emptying a type sII clathrate hydrate. Nature, 516(7530), 231–233. doi: 10.1038/nature14014. Guo DD, Ning FL, Ou WJ, Zhang L, He ZJ, Fang B. 2019. Research progress of nanoparticles and hydrate formation. Geological Science and Technology Information, 38(6), 102–118. doi: 10.19509/j.cnki.dzkq.2019.0612. Guo GJ, Zhang YG, Zhao YJ, Refson K, Shan GH. 2004. Lifetimes of cagelike water clusters immersed in bulk liquid water: A molecular dynamics study on gas hydrate nucleation mechanisms. Journal of Chemical Physics, 121(3), 1542–1547. doi: 10.1063/1.1763888. Guo GJ, Zhang YG, Refson K. 2005. Effect of H-bond topology on the lifetimes of cagelike water clusters immersed in liquid water and the probability distribution of these lifetimes: Implications for hydrate nucleation mechanisms. Chemical Physics Letters, 413, 415–419. doi: 10.1016/j.cplett.2005.08.015. Guo GJ, Zhang YG, Liu H. 2007. Effect of methane adsorption on the lifetime of a dodecahedral water cluster immersed in liquid water: A molecular dynamics study on the hydrate nucleation mechanisms. The Journal of Physical Chemistry C, 111(6), 2595–2606. doi: 10.1021/jp064271t. Guo GJ, Li M, Zhang YG, Wu CH. 2009. Why can water cages adsorb aqueous methane? A potential of mean force calculation on hydrate nucleation mechanisms. Physical Chemistry Chemical Physics, 11(44), 10427–10437. doi: 10.1039/B913898F. Guo GJ, Zhang YG, Liu CJ, Li KH. 2011. Using the face-saturated incomplete cage analysis to quantify the cage compositions and cage linking structures of amorphous phase hydrates. Physical Chemistry Chemical Physics, 13(25), 12048–12057. doi: 10.1039/c1cp20070d. Guo GJ, Rodger PM. 2013. Solubility of aqueous methane under metastable conditions: implications for gas hydrate nucleation. The Journal of Physical Chemistry B, 117(21), 6498–6504. doi: 10.1021/jp3117215. Guo GJ, Zhang ZC. 2021. Open questions on methane hydrate nucleation. Communications Chemistry, 4, 102. doi: 10.1038/s42004-021-00539-6. Hall KW, Carpendale S, Kusalik PG. 2016. Evidence from mixed hydrate nucleation for a funnel model of crystallization. Proceedings of the National Academy of Sciences of the United States of America, 113(43), 12041–12046. doi: 10.1073/pnas.1610437113. Hassanpouryouzband A, Joonaki E, Vasheghani Farahani M, Takeya S, Ruppel C, Yang J, English NJ, Schicks JM, Edlmann K, Mehrabian H, Aman ZM, Tohidi B. 2020. Gas hydrates in sustainable chemistry. Chemical Society Reviews, 49(15), 5225–5309. doi: 10.1039/c8cs00989a. Haugan PM, Drange H. 1992. Sequestration of CO2 in the deep ocean by shallow injection. Nature, 357(6376), 318–320. doi: 10.1038/357318a0. Hawtin RW, Quigley D, Rodger PM. 2008. Gas hydrate nucleation and cage formation at a water/methane interface. Physical Chemistry Chemical Physics, 10(32), 4853–4864. doi: 10.1039/B807455k. He ZJ, Gupta KM, Linga P, Jiang JW. 2016. Molecular insights into the nucleation and growth of CH4 and CO2 mixed hydrates from microsecond simulations. The Journal of Physical Chemistry C, 120(44), 25225–25236. doi: 10.1021/acs.jpcc.6b07780. He ZJ, Linga P, Jiang JW. 2017a. CH4 hydrate formation between silica and graphite surfaces: insights from microsecond molecular dynamics simulations. Langmuir, 33(43), 11956–11967. doi: 10.1021/acs.langmuir.7b02711. He ZJ, Linga P, Jiang JW. 2017b. What are the key factors governing the nucleation of CO2 hydrate? Physical Chemistry Chemical Physics, 19(24), 15657–15661. doi: 10.1039/c7cp01350g He ZJ, Mi FY, Ning FL. 2021. Molecular insights into CO2 hydrate formation in the presence of hydrophilic and hydrophobic solid surfaces. Energy, 234, 121260. doi: 10.1016/j.energy.2021.121260. Hester KC, Brewer PG. 2009. Clathrate hydrates in nature. Annual Review of Marine Science, 1, 303–327. doi: 10.1146/annurev.marine.010908.163824. Hirai S, Okazaki K, Kuraoka S, Katsuyuki K. 1997. Molecular dynamics simulation for the formation of argon clathrate-hydrate structure. Microscale Thermophysical Engineering, 1(4), 293–301. doi: 10.1080/108939597200151. Hu WF, Chen C, Sun JY, Zhang N, Zhao JF, Liu Y, Ling Z, Li WZ, Liu WG, Song YC. 2022. Three-body aggregation of guest molecules as a key step in methane hydrate nucleation and growth. Communications Chemistry, 5, 33. doi: 10.1038/s42004-022-00652-0. Jacobson LC, Hujo W, Molinero V. 2010a. Nucleation pathways of clathrate hydrates: effect of guest size and solubility. The Journal of Physical Chemistry B, 114(43), 13796–13807. doi: 10.1021/jp107269q. Jacobson LC, Hujo W, Molinero V. 2010b. Amorphous precursors in the nucleation of clathrate hydrates. Journal of the American Chemical Society, 132(33), 11806–11811. doi: 10.1021/ja1051445. Ji HQ, Chen DY, Wu GZ. 2017. Molecular mechanisms for cyclodextrin-promoted methane hydrate formation in water. The Journal of Physical Chemistry C, 121(38), 20967–20975. doi: 10.1021/acs.jpcc.7b03338. Jiménez-Ángeles F, Firoozabadi A. 2014. Nucleation of methane hydrates at moderate subcooling by molecular dynamics simulations. The Journal of Physical Chemistry C, 118(21), 11310–11318. doi: 10.1021/jp5002012. Jiménez-Ángeles F, Firoozabadi A. 2015. Enhanced hydrate nucleation near the limit of stability. The Journal of Physical Chemistry C, 119(16), 8798–8804. doi: 10.1021/acs.jpca.5b01869. Kamata S, Nimmo F, Sekine Y, Kuramoto K, Noguchi N, Kimura J, Tani A. 2019. Pluto’s ocean is capped and insulated by gas hydrates. Nature Geoscience, 12(6), 407–410. doi: 10.1038/s41561-019-0369-8. Knott BC, Molinero V, Doherty MF, Peters B. 2012. Homogeneous nucleation of methane hydrates: unrealistic under realistic conditions. Journal of the American Chemical Society, 134(48), 19544–19547. doi: 10.1021/ja309117d. Koh CA, Sloan ED, Sum AK, Wu DT. 2011. Fundamentals and applications of gas hydrates. Annual Review of Chemical and Biomolecular Engineering 2, 237–257. doi: 10.1146/annurev-chembioeng-061010-114152. Kvenvolden KA. 1993. Gas hydrates—geological perspective and global change. Reviews of Geophysics, 31(2), 173–187. doi: 10.1029/93RG00268. Kvenvolden KA. 1999. Potential effects of gas hydrate on human welfare. Proceedings of the National Academy of Sciences of the United States of America, 96(7), 3420–3426. doi: 10.1073/pnas.96.7.3420. Lauricella M, Meloni S, English NJ, Peters B, Ciccotti G. 2014. Methane clathrate hydrate nucleation mechanism by advanced molecular simulations. The Journal of Physical Chemistry C, 118(40), 22847–22857. doi: 10.1021/jp5052479. Lauricella M, Meloni S, Liang S, English NJ, Kusalik PG, Ciccotti G. 2015. Clathrate structure-type recognition: Application to hydrate nucleation and crystallisation. Journal of Chemical Physics, 142(24), 244503. doi: 10.1063/1.4922696. Lauricella M, Ciccotti G, English NJ, Peters B, Meloni S. 2017. Mechanisms and nucleation rate of methane hydrate by dynamical nonequilibrium molecular dynamics. The Journal of Physical Chemistry C, 121(43), 24223–24234. doi: 10.1021/acs.jpcc.7b05754. Lei L, Seol Y. 2019. High-saturation gas hydrate reservoirs—A pore scale investigation of their formation from free gas and dissociation in sediments. Journal of Geophysical Research:Solid Earth, 124(12), 12430–12444. doi: 10.1029/2019JB018243. Li JF, Ye JL, Qin XW, Qiu HJ, Wu NY, Lu HL, Xie WW, Lu JA, Peng F, Xu ZQ, Lu C, Kuang ZG, Wei JG, Liang QY, Lu HF, Kou BB. 2018. The first offshore natural gas hydrate production test in South China Sea. China Geology, 1(1), 5–16. doi: 10.31035/cg2018003. Li LW, Zhong J, Yan YG, Zhang J, Xu JF, Francisco JS, Zeng XC. 2020. Unraveling nucleation pathway in methane clathrate formation. Proceedings of the National Academy of Sciences of the United States of America, 117(40), 24701–24708. doi: 10.1073/pnas.2011755117. Liang RD, Xu HJ, Shen YN, Sun SM, Xu JY, Meng S, Shen YR, Tian CS. 2019. Nucleation and dissociation of methane clathrate embryo at the gas-water interface. Proceedings of the National Academy of Sciences of the United States of America, 116(47), 23410–23415. doi: 10.1073/pnas.1912592116. Liang S, Kusalik PG. 2010. Explorations of gas hydrate crystal growth by molecular simulations. Chemical Physics Letters, 494(4-6), 123–133. doi: 10.1016/j.cplett.2010.05.088. Liang S, Kusalik PG. 2011a. The mobility of water molecules through gas hydrates. Journal of the American Chemical Society, 133(6), 1870–1876. doi: 10.1021/ja108434h. Liang S, Kusalik PG. 2011b. Exploring nucleation of H2S hydrates. Chemical Science, 2(7), 1286–1292. doi: 10.1039/c1sc00021g. Liang S, Kusalik PG. 2013. Nucleation of gas hydrates within constant energy systems. The Journal of Physical Chemistry B, 117(5), 1403–1410. doi: 10.1021/jp308395x. Liang S, Kusalik PG. 2015a. The nucleation of gas hydrates near silica surfaces. Canadian Journal of Chemistry, 93(8), 791–798. doi: 10.1139/cjc-2014-0443. Liang S, Kusalik PG. 2015b. Communication: structural interconversions between principal clathrate hydrate structures. Journal of Chemical Physics, 143(1), 011102. doi: 10.1063/1.4923465. Liang S, Hall KW, Laaksonen A, Zhang ZC, Kusalik PG. 2019. Characterizing key features in the formation of ice and gas hydrate systems. Philosophical Transactions Mathematical Physical & Engineering Sciences, 377(2146), 20180167. doi: 10.1098/rsta.2018.0167. Lin ZY, Wu DT, Lin ST. 2018. Equilibrium and transport properties of methane at the methane/water interface with the presence of SDS. The Journal of Physical Chemistry C, 122(51), 29259–29267. doi: 10.1021/acs.jpcc.8b08830. Liu CJ, Zhang ZC, Guo GJ. 2016. Effect of guests on the adsorption interaction between a hydrate cage and guests. RSC Advances, 6(108), 106443–106452. doi: 10.1039/c6ra21513k. Loveday JS, Nelmes RJ, Guthrie M, Belmonte SA, Allan DR, Klug DD, Tse JS, Handa YP. 2001. Stable methane hydrate above 2 GPa and the source of Titan's atmospheric methane. Nature, 410(6829), 661–663. doi: 10.1038/35070513. Matsumoto M. 2010. Four-Body Cooperativity in Hydrophonic Association of Methane. Journal of Physical Chemistry Letters, 1(10), 1552–1556. doi: 10.1021/jz100340e. Mehrabian H, Bellucci MA, Walsh MR, Trout BL. 2018. Effect of salt on antiagglomerant surface adsorption in natural gas hydrates. The Journal of Physical Chemistry C, 122(24), 12839–12849. doi: 10.1021/acs.jpcc.8b03154. Mehrabian H, Walsh MR, Trout BL. 2019. In silico analysis of the effect of alkyl tail length on antiagglomerant adsorption to natural gas hydrates in brine. The Journal of Physical Chemistry C, 123(28), 17239–17248. doi: 10.1021/acs.jpcc.9b01952. Mehrabian H, Trout BL. 2020. In silico engineering of hydrate anti-agglomerant molecules using bias-exchange metadynamics simulations. The Journal of Physical Chemistry C, 124(35), 18983–18992. doi: 10.1021/acs.jpcc.0c03251. Michalis VK, Costandy J, Tsimpanogiannis IN, Stubos AK, Economou IG. 2015. Prediction of the phase equilibria of methane hydrates using the direct phase coexistence methodology. Journal of Chemical Physics, 142(4), 044501. doi: 10.1063/1.4905572. Mohr S, Hoevelmann F, Wylde J, Schelero N, Sarria J, Purkayastha N, Ward Z, Navarro Acero P, Michalis VK. 2021. Ranking the efficiency of gas hydrate anti-agglomerants through molecular dynamic simulations. The Journal of Physical Chemistry B, 125(5), 1487–1502. doi: 10.1021/acs.jpcb.0c08969. Momma K, Izumi F. 2011. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. Journal of Applied Crystallography, 44(6), 1272–1276. doi: 10.1107/s0021889811038970. Moon C, Taylor PC, Rodger PM. 2003. Molecular dynamics study of gas hydrate formation. Journal of the American Chemical Society, 125(16), 4706–4707. doi: 10.1021/ja028537v. Mueller T, Hernandez A, Wang C. 2020. Machine learning for interatomic potential models. Journal of Chemical Physics, 152(5), 050902. doi: 10.1063/1.5126336. Naullage PM, Bertolazzo AA, and Molinero V. 2019. How do surfactants control the agglomeration of clathrate hydrates? ACS Central Science, 5(3), 428–439. doi: 10.1021/acscentsci.8b00755 Pirzadeh P, Beaudoin EN, Kusalik PG. 2012. Interfacial free energy: An entropy portent to energy changes. Crystal Growth & Design, 12(1), 124–128. doi: 10.1021/cg200861e. Pirzadeh P, Kusalik PG. 2013. Molecular insights into clathrate hydrate nucleation at an ice-solution interface. Journal of the American Chemical Society, 135(19), 7278–7287. doi: 10.1021/ja400521e. Qin XW, Lu JA, Lu HL, Qiu HJ, Liang JQ, Kang DJ, Zhan LS, Lu HF, Kuang ZG. 2020. Coexistence of natural gas hydrate, free gas and water in the reservoir system in the Shenhu Area, South China Sea. China Geology, 2, 1–11. doi: 10.31035/cg2020038. Qin XW, Liang QY, Ye JL, Yang L, Qiu HJ, Xie WW, Liang JQ, Lu JA, Lu C, Lu HL, Ma BJ, Kuang ZG, Wei JG, Lu HF, Kou BB. 2020. The response of temperature and pressure of hydrate reservoirs in the first gas hydrate production test in South China Sea. Applied Energy, 278, 115649. doi: 10.1016/j.apenergy.2020.115649. Radhakrishnan R, Trout BL. 2002. A new approach for studying nucleation phenomena using molecular simulations: Application to CO2 hydrate clathrates. Journal of Chemical Physics, 117(4), 1786–1796. doi: 10.1063/1.1485962. Ripmeester JA, Alavi S. 2016. Some current challenges in clathrate hydrate science: Nucleation, decomposition and the memory effect. Current Opinion in Solid State & Materials Science, 20(6), 344–351. doi: 10.1016/j.cossms.2016.03.005. Rodger PM, Forester TR, and Smith W. 1996. Simulations of the methane hydrate methane gas interface near hydrate forming conditions. Fluid Phase Equilibria, 116(1–2), 326–332. doi: 10.1016/0378-3812(95)02903-6. Rozmanov D, Kusalik PG. 2011. Temperature dependence of crystal growth of hexagonal ice (Ih). Physical Chemistry Chemical Physics, 13(34), 15501–15511. doi: 10.1039/c1cp21210a. Sarupria S, Debenedetti PG. 2012. Homogeneous nucleation of methane hydrate in microsecond molecular dynamics simulations. The Journal of Physical Chemistry Letters, 3(20), 2942–2947. doi: 10.1021/jz3012113. Shi BH, Song SF, Chen YC, Duan X, Liao QY, Fu SK, Liu LH, Sui JH, Jia JP, Liu HT, Zhu YM, Song CX, Lin DC, Wang T, Wang JA, Yao HY, Gong J. 2021. Status of natural gas hydrate flow assurance research in China: A review. Energy & Fuels, 35(5), 3611–3658. doi: 10.1021/acs.energyfuels.0c04209. Sloan ED, Fleyfel F. 1991. A molecular mechanism for gas hydrate nucleation from ice. AIChE Journal, 37(9), 1281–1292. doi: 10.1002/aic.690370902. Sloan ED. 2003. Fundamental principles and applications of natural gas hydrates. Nature, 426(6964), 353–363. doi: 10.1038/nature02135. Sloan ED, Koh CA. 2008. Clathrate hydrates of natural gasses. CRC Press, Boca Raton, FL, doi: 10.1201/9781420008494 Sosso GC, Chen J, Cox SJ, Fitzner M, Pedevilla P, Zen A, Michaelides A. 2016. Crystal nucleation in liquids: Open questions and future challenges in molecular dynamics simulations. Chemical Reviews, 116(12), 7078–7116. doi: 10.1021/acs.chemrev.5b00744. Sum AK. 2013. Prevention, management, and remediation approaches for gas hydrates in the flow assurance of oil/gas flowlines, in Proceedings OTC Brasil 2013, Volume All Days: OTC-24396-MS. Tang LL, Su Y, Liu Y, Zhao JJ, Qiu RF. 2012. Nonstandard cages in the formation process of methane clathrate: stability, structure, and spectroscopic implications from first-principles. Journal of Chemical Physics, 136(22), 224508. doi: 10.1063/1.4728157. Tian HQ, Zhang ZC. 2020. Revealing the growth of H2 + THF binary hydrate through molecular simulations. Energy & Fuels, 34(11), 15004–15010. doi: 10.1021/acs.energyfuels.0c03096. Tréhu AM, Ruppel C, Holland M, Dickens GR, Torres ME, Collett TS, Goldberg D, Riedel R, Schultheiss P. 2006. Gas hydrates in marine sediments - lessons from scientific ocean drilling. Oceanography, 19(124), 124–142. doi: 10.5670/oceanog.2006.11. Tung YT, Chen LJ, Chen YP, Lin ST. 2010. The growth of structure I methane hydrate from molecular dynamics simulations. The Journal of Physical Chemistry B, 114(33), 10804–10813. doi: 10.1021/jp102874s. Tung YT, Chen LJ, Chen YP, Lin ST. 2011. Growth of structure i carbon dioxide hydrate from molecular dynamics simulations. The Journal of Physical Chemistry C, 115(15), 7504–7515. doi: 10.1021/jp12205x. Tung YT, Chen LJ, Chen YP, Lin ST. 2012. Molecular dynamics study on the growth of structure I methane hydrate in aqueous solution of sodium chloride. The Journal of Physical Chemistry B, 116(48), 14115–14125. doi: 10.1021/jp308224v. Vatamanu J, Kusalik PG. 2006a. Unusual crystalline and polycrystalline structures in methane hydrates. Journal of the American Chemical Society, 128(49), 15588–15589. doi: 10.1021/ja066515t. Vatamanu J, Kusalik PG. 2006b. Molecular insights into the heterogeneous crystal growth of sI methane hydrate. The Journal of Physical Chemistry B, 110(32), 15896–15904. doi: 10.1021/jp0616841. Vatamanu J, Kusalik PG. 2008. Heterogeneous crystal growth of methane hydrate on its sII [001] crystallographic face. The Journal of Physical Chemistry B, 112(8), 2399–2404. doi: 10.1021/jp077583k. Vatamanu J, Kusalik PG. 2010. Observation of two-step nucleation in methane hydrates. Physical Chemistry Chemical Physics, 12(45), 15065–15072. doi: 10.1039/C0cp00551g. Walsh MR, Koh CA, Sloan ED, Sum AK, Wu DT. 2009. Microsecond simulations of spontaneous methane hydrate nucleation and growth. Science, 326(5956), 1095–1098. doi: 10.1126/science.1174010. Walsh MR, Beckham GT, Koh CA, Sloan ED, Wu DT, Sum AK. 2011a. Methane hydrate nucleation rates from molecular dynamics simulations: Effects of aqueous methane concentration, interfacial curvature, and system size. The Journal of Physical Chemistry C, 115(43), 21241–21248. doi: 10.1021/jp206483q. Walsh MR, Rainey JD, Lafond PG, Park DH, Beckham GT, Jones MD, Lee KH, Koh CA, Sloan ED, Wu DT, Sum AK. 2011b. The cages, dynamics, and structuring of incipient methane clathrate hydrates. Physical Chemistry Chemical Physics, 13(44), 19951–19959. doi: 10.1039/C1cp21899a. Wang PW, Wu DT, Lin ST. 2021. Promotion mechanism for the growth of CO2 hydrate with urea using molecular dynamics simulations. Chemical Communications, 57(43), 5330–5333. doi: 10.1039/d0cc06165d. Wang R, Liu TL, Ning FL, Ou WJ, Zhang L, Wang Z, Peng L, Sun JX, Liu ZC, Li TS, Sun HC, Jiang GS. 2018. Effect of hydrophilic silica nanoparticles on hydrate formation: Insight from the experimental study. Journal of Energy Chemistry, 30, 90–100. doi: 10.1016/j.jechem.2018.02.021. Wilson DT, Barnes BC, Wu DT, Sum AK. 2016. Molecular dynamics simulations of the formation of ethane clathrate hydrates. Fluid Phase Equilibria, 413, 229–234. doi: 10.1016/j.fluid.2015.12.001. Wu JY, Chen LJ, Chen YP, Lin ST. 2016. Molecular dynamics study on the nucleation of methane + tetrahydrofuran mixed guest hydrate. Physical Chemistry Chemical Physics, 18(15), 9935–9947. doi: 10.1039/c5cp06419h. Wu NY, Li YL, Wan YZ, Sun JY, Huang L, Mao PX. 2021. Prospect of marine natural gas hydrate stimulation theory and technology system. Natural Gas Industry B, 8(2), 173–187. doi: 10.1016/j.ngib.2020.08.003. Xu JF, Li LW, Liu JX, Wang XP, Yan YG, Zhang J. 2018. The molecular mechanism of the inhibition effects of PVCaps on the growth of sI hydrate: an unstable adsorption mechanism. Physical Chemistry Chemical Physics, 20(12), 8326–8332. doi: 10.1039/c8cp00010g. Yagasaki T, Matsumoto M, Andoh Y, Okazaki S, Tanaka H. 2014a. Dissociation of methane hydrate in aqueous NaCl solutions. The Journal of Physical Chemistry B, 118(40), 11797–11804. doi: 10.1021/jp507978u. Yagasaki T, Matsumoto M, Andoh Y, Okazaki S, Tanaka H. 2014b. Effect of bubble formation on the dissociation of methane hydrate in water: a molecular dynamics study. The Journal of Physical Chemistry B, 118(7), 1900–1906. doi: 10.1021/jp412692d. Yagasaki T, Matsumoto M, Tanaka H. 2015. Adsorption mechanism of inhibitor and guest molecules on the surface of gas hydrates. Journal of the American Chemical Society, 137(37), 12079–12085. doi: 10.1021/jacs.5b07417. Yagasaki T, Matsumoto M, Tanaka H. 2016a. Mechanism of slow crystal growth of tetrahydrofuran clathrate hydrate. The Journal of Physical Chemistry C, 120(6), 3305–3313. doi: 10.1021/acs.jpcc.5b10293. Yagasaki T, Matsumoto M, Tanaka H. 2016b. Formation of clathrate hydrates of water-soluble guest molecules. The Journal of Physical Chemistry C, 120(38), 21512–21521. doi: 10.1021/acs.jpcc.6b06498. Yagasaki T, Matsumoto M, Tanaka H. 2018a. Molecular dynamics study of kinetic hydrate inhibitors: the optimal inhibitor size and effect of guest species. The Journal of Physical Chemistry C, 123(3), 1806–1816. doi: 10.1021/acs.jpcc.8b09834. Yagasaki T, Matsumoto M, Tanaka H. 2018b. Adsorption of kinetic hydrate inhibitors on growing surfaces: a molecular dynamics study. The Journal of Physical Chemistry B, 122(13), 3396–3406. doi: 10.1021/acs.jpcb.7b10356. Yan KF, Li XS, Chen ZY, Xia ZM, Xu CG, Zhang ZQ. 2016. Molecular dynamics simulation of the crystal nucleation and growth behavior of methane hydrate in the presence of the surface and nanopores of porous sediment. Langmuir, 32(31), 7975–7984. doi: 10.1021/acs.langmuir.6b01601. Yang MJ, Zhao J, Zheng J-N, Song YC. 2019. Hydrate reformation characteristics in natural gas hydrate dissociation process: A review. Applied Energy, 256, 113878. doi: 10.1016/j.apenergy.2019.113878. Ye JL, Qin XW, Xie WW, Lu HL, Ma BJ, Qiu HJ, Liang GQ, Lu GA, Kuang ZG, Lu C, Liang QY, Wei SP, Yu YJ, Liu CH, Li B, Shen KX, Shi HX, Lu QP, Li J, Kou BB, Song G, Li B, Zhang HE, Lu HF, Ma C, Dong YF, Bian H. 2020. The second natural gas hydrate production test in the South China Sea. China Geology, 47(3), 197–209. doi: 10.31035/cg2020043. Ye YG, Zhang J, Xi SB, Liu CL, Ren SH, Sui WD. 2003. Experimental technique for marine gas hydrates. Marine Geology & Quaternary Geology, 23(1), 119–123. You K, Flemings PB, Malinverno A, Collett TS, Darnell K. 2019. Mechanisms of methane hydrate formation in geological systems. Reviews of Geophysics, 57(4), 1146–1196. doi: 10.1029/2018RG000638. Yuhara D, Barnes BC, Suh D, Knott BC, Beckham GT, Yasuoka K, Wu DT, Sum AK. 2015. Nucleation rate analysis of methane hydrate from molecular dynamics simulations. Faraday Discussions, 179, 463–474. doi: 10.1039/c4fd00219a. Zhang YC, Liu LL, Wang DG, Zhang Z, Li CF, Meng QG, Liu CL. 2021a. The interface evolution during methane hydrate dissociation within quartz sands and its implications to the permeability prediction based on NMR data. Marine and Petroleum Geology, 129, 105065. doi: 10.1016/j.marpetgeo.2021.105065. Zhang YC, Wan YZ, Liu LL, Wang DG, Li CF, Liu CL, Wu NY. 2021b. Changes in reaction surface during the methane hydrate dissociation and its implications for hydrate production. Energy, 230, 120848. doi: 10.1016/j.energy.2021.120848. Zhang ZC, Walsh MR, Guo GJ. 2015. Microcanonical molecular simulations of methane hydrate nucleation and growth: evidence that direct nucleation to sI hydrate is among the multiple nucleation pathways. Physical Chemistry Chemical Physics, 17(14), 8870–8876. doi: 10.1039/c5cp00098j. Zhang ZC, Liu CJ, Walsh MR, Guo GJ. 2016. Effects of ensembles on methane hydrate nucleation kinetics. Physical Chemistry Chemical Physics, 18(23), 15602–15608. doi: 10.1039/c6cp02171a. Zhang ZC, Guo GJ. 2017. The effects of ice on methane hydrate nucleation: a microcanonical molecular dynamics study. Physical Chemistry Chemical Physics, 19(29), 19496–19505. doi: 10.1039/c7cp03649c. Zhang ZC, Kusalik PG, Guo GJ. 2018a. Bridging solution properties to gas hydrate nucleation through guest dynamics. Physical Chemistry Chemical Physics, 20(38), 24535–24538. doi: 10.1039/c8cp04466j. Zhang ZC, Kusalik PG, Guo GJ. 2018b. Molecular insight into the growth of hydrogen and methane binary hydrates. The Journal of Physical Chemistry C, 122(14), 7771–7778. doi: 10.1021/acs.jpcc.8b00842. Zhang ZC, Kusalik PG, Guo GJ. 2019. Might a 2, 2-dimethylbutane molecule serve as a site to promote gas hydrate nucleation? The Journal of Physical Chemistry C, 123(33), 20579–20586. doi: 10.1021/acs.jpcc.9b04518. Zhang ZC, Guo GJ, Wu NY, Kusalik PG. 2020. Molecular insights into guest and composition dependence of mixed hydrate nucleation. The Journal of Physical Chemistry C, 124(45), 25078–25086. doi: 10.1021/acs.jpcc.0c07375. Zhang ZC, Kusalik PG, Guo GJ, Ning FL, Wu NY. 2021. Insight on the stability of polycrystalline natural gas hydrates by molecular dynamics simulations. Fuel, 289, 119946. doi: 10.1016/j.fuel.2020.119946. Zheng JJ, Chong ZR, Qureshi MF, Linga P. 2020. Carbon dioxide sequestration via gas hydrates: A potential pathway toward decarbonization. Energy & Fuels, 34(9), 10529–10546. doi: 10.1021/acs.energyfuels.0c02309. -
Access History
-
Figure 1.
Cages and three common crystal structures of NGHs. Structures were generated with VESTA-3 (Momma K and Izumi F, 2011).
-
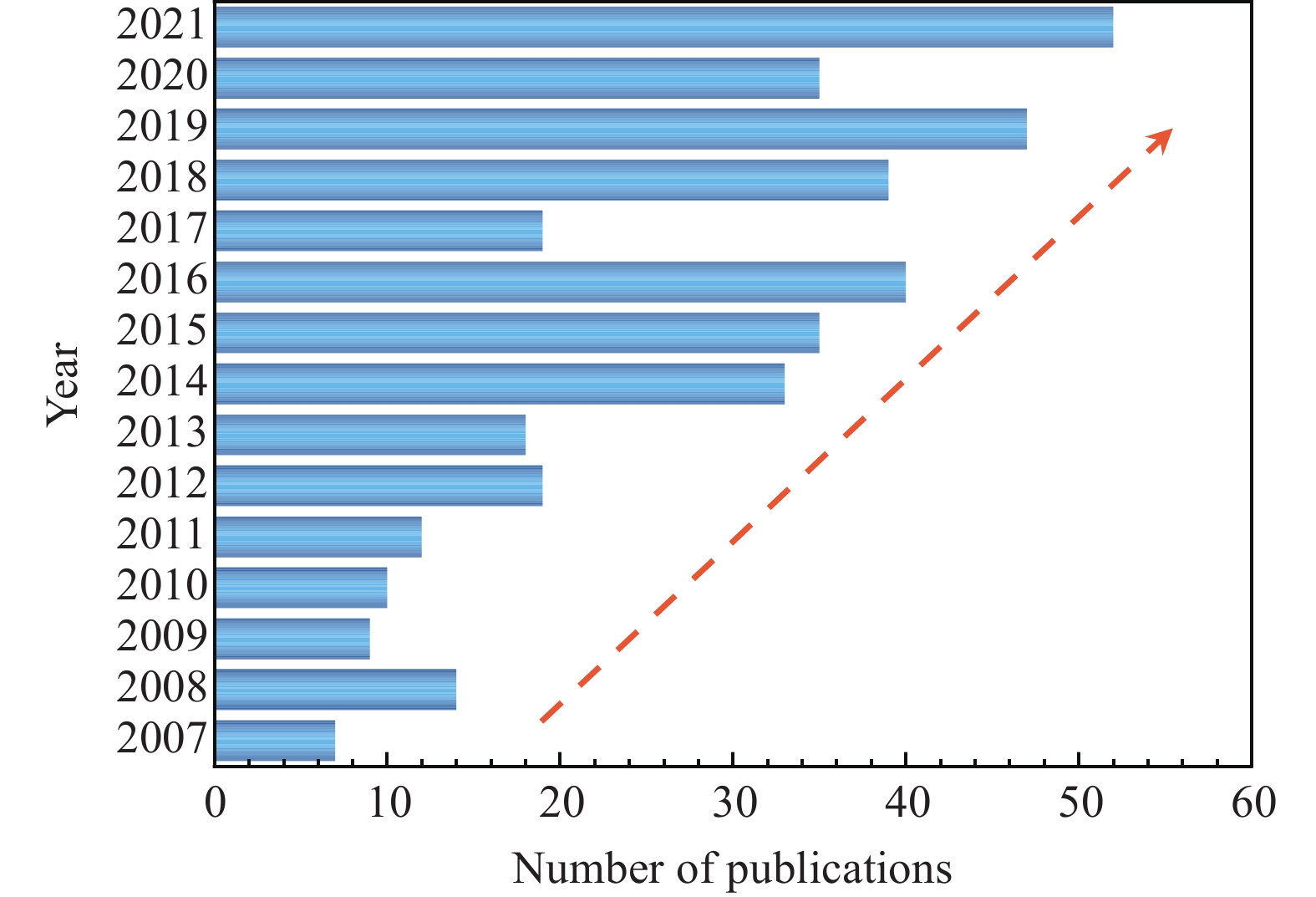
Figure 2.
The number of publications from the past 15 years on molecular simulation studies of gas hydrate formation. Data is from the Web of Sciences with keywords of “gas hydrate formation” and “molecular simulation”.
-
Figure 3.
Schematic model of LCH proposed by Sloan and co-workers (after Sloan ED and Fleyfel F, 1991; Christiansen RL and Sloan ED, 1994).
-
Figure 4.
Schematic model showing the local structuring hypothesis (LSH). Six snapshots along nucleation are depicted. Carbon dioxide molecules are in cyan. White lines mean hydrogen bonds between water molecules. Modified with permission from Radhakrishnan R and Trout BL (2002).
-
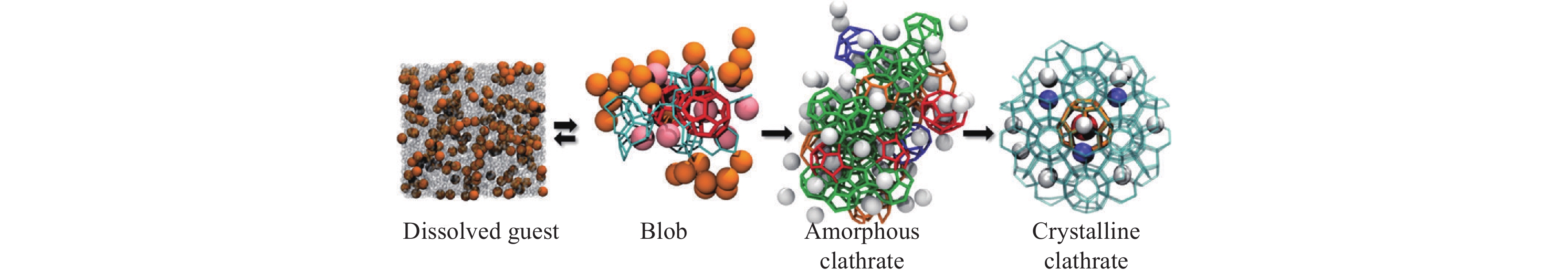
Figure 5.
The cage adsorption hypothesis (CAH). When the guest concentration reaches the critical value (for methane, corresponding to the average separation between them being 0.88 nm), the first spontaneous formed cage will adsorb the neighboring guests (a), then water molecules will rearrange to form more cages and more guests will diffuse toward the cage cluster (b). This process will go on and when the size of cage cluster is large enough, hydrate grows inevitably (c). The potential mean force (PMF) between 512 cage and methane molecule (d).
-
Figure 6.
Snapshots, potential energy, and order parameter showing the first spontaneous formation trajectory of methane hydrate from microsecond molecular simulations at 250 K and 50 MPa. Methane is represented as blue spheres, and hydrogen bonds between water molecules in red lines. The figure reproduced with permission from Walsh MR et al. (2009).
-
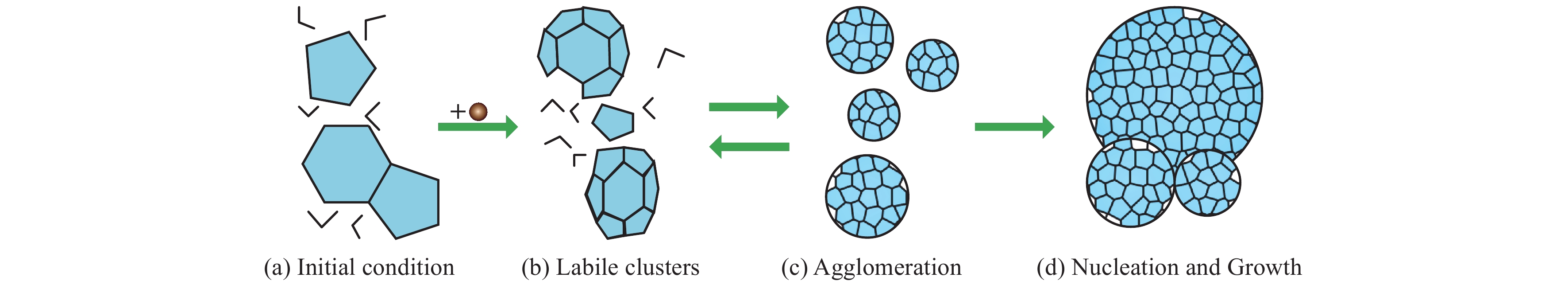
Figure 7.
Schematic for the two-step mechanism as proposed by Molinero’s group. The figure is reproduced with permission from Jacobson LC et al. (2010b).
-
Figure 8.
Snapshots from one of the nucleation trajectories of methane hydrate, where direct nucleation of crystalline hydrate was observed. The figure was reproduced with permission from Zhang ZC et al. (2015).
-
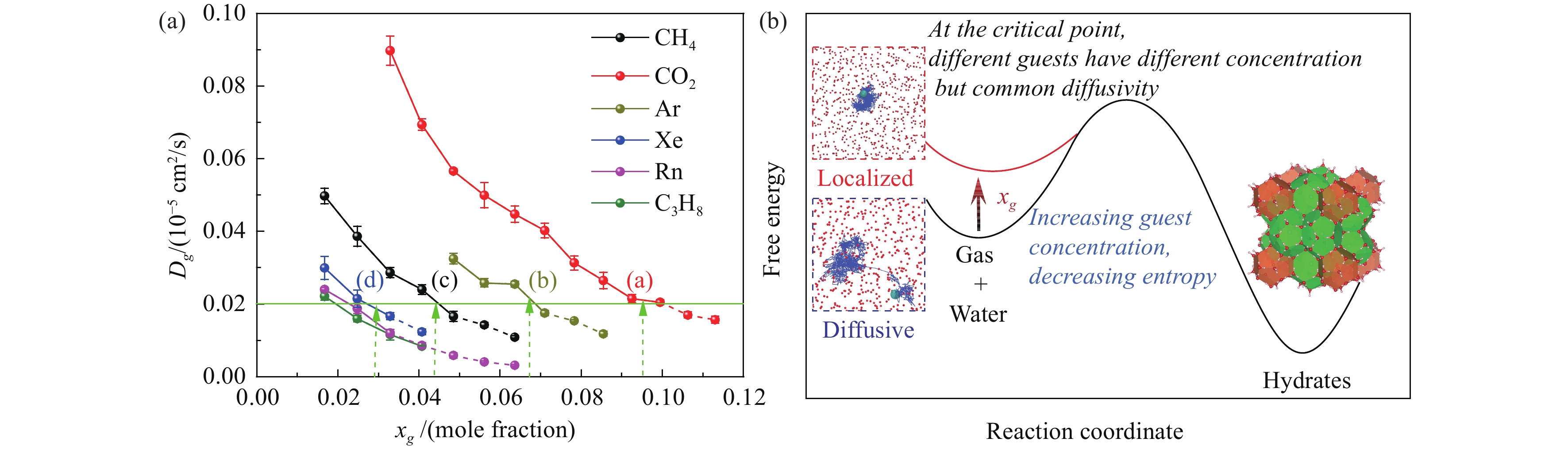
Figure 9.
(a) Critical self-diffusivity for gas hydrate nucleation and (b) schematic for thermodynamic of hydrate nucleation. Figures reprinted with permission from Zhang ZC et al. (2018a).
-
Figure 10.
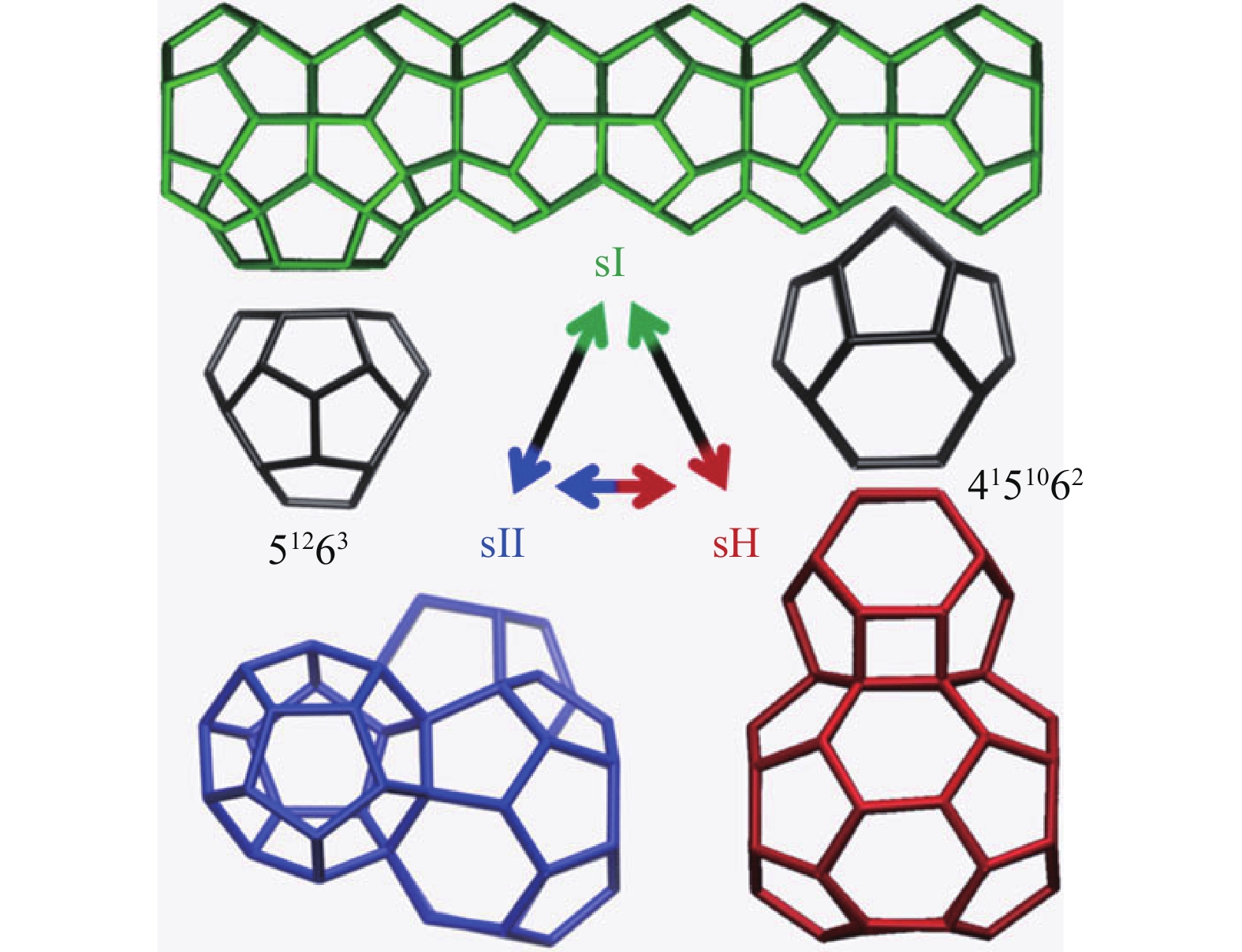
Possible structural interconversion between principal gas hydrate structures sI, sII, and sH. Reprinted with permission from Liang S and Kusalik PG (2015b).
-
Figure 11.
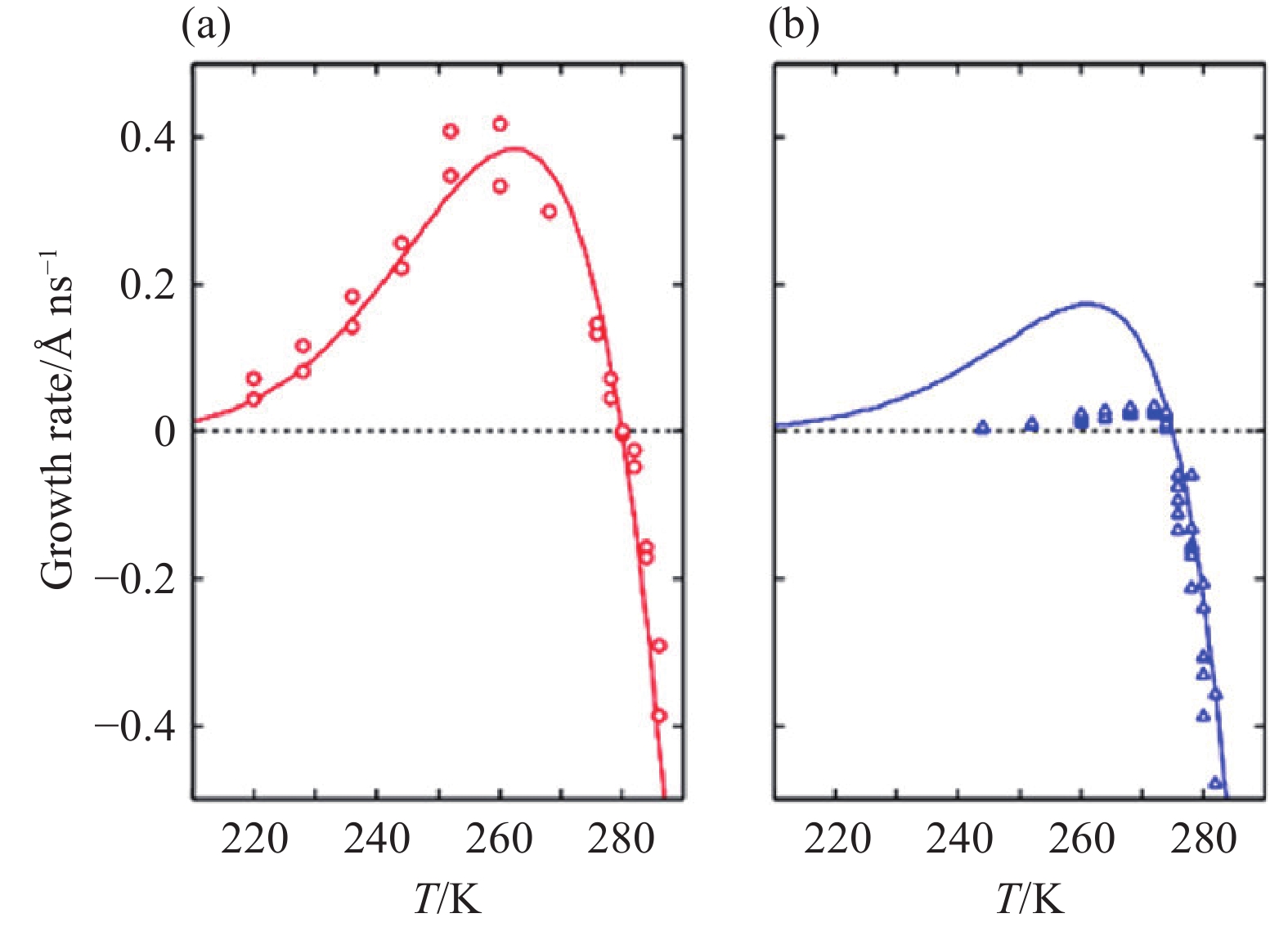
Growth rates of (a) ethylene oxide hydrate and (b) THF hydrate. Reprinted with permission from Yagasaki T et al. (2016b).
-
Figure 12.
Schematic of the energy landscape for hydrate nucleation. The system’s potential energy dictates the depth of the point in the funnel, and the funnel’s width at a point is the system’s entropy in (c). Reprinted with permission from Liang S et al. (2019).
-
Figure 13.
Schematic of methane hydrate nucleation. Reprinted with permission from Guo GJ and Zhang ZC (2021).





 DownLoad:
DownLoad: